Sample Content
GPAT Study Material - Medicinal Chemistry : Local Anaesthetics
It is impossible to provide effective dental care or in minor surgeries, without the use of local anesthetics. This drug class has an impressive history of safety and efficacy. A Local anesthetic agent is a drug, that, when given either topically or parenterally to a localized area, produces a state of local anesthesia by reversibly blocking the nerve conductances. Local anesthetics interrupt neural conduction by inhibiting the influx of sodium ions. In most cases, this follows their diffusion through the neural membrane into the axoplasm, where they enter sodium channels and prevent them from assuming an active or open state. The local anesthetic molecule consists of 3 components:(a) lipophilic aromatic ring, (b) amide chain or intermediate ester, and (c) terminal amine. Each of these gives the distinct properties to the molecule. Ref the following figure:
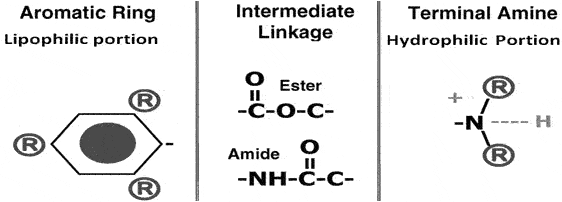
Structure-Activity Relationships: Most of the clinically useful local anesthetics are tertiary amines with pKa values of 7.5 to 9.0. These drugs exhibit their local anesthetics properties by binding of the onium ions to a selective site within the sodium channels. For this reason, any structural modifications that alter the lipid solubility, pKa, & metabolic in activation definitely have a pronounced effect on its local anesthetic properties.
Lipophilic Portion-of the molecular is essential for local anesthetics activity. This portion of the molecule consists of either an aromatic group- directly attached to a carbonyl function (amino ester series) a 2,6-dimethyl phenyl group, attached to a carbonyl function through an –NH-group(amino amide series). Both of these group highly lipophilic play an important role in the binding of local anesthetics to the channel receptor.
In the amino ester series, an electron donating substituent in the ortho or Para or both positions increases local anesthetic potency. Such group as an amino (procaine,butacaine) an alkylamino (tetracaine), or an alkoxy (propoxycainecan) contribute electron density to the aromatic ring by both resonance & inductive effects, thereby enhancing local anesthetic potency. It is generally accepted that local anesthetics having higher lipid solubility & lower pKa values appear to exhibit more rapid onset and lower toxicity.
Electron with drawing groups such as nitro (-NO2 ), carbonyl (-CO-), and nitrile (-CN) reduced the local anesthetic activity. The shorter duration of action, observed with chloroprocaine explained by the inductive effect of the o-chloro group, which pulls the electron density. When an amino or an alkoxy group is attached to the meta position of the aromatic ring, however, no resonance delocalization of their electrons is possible.
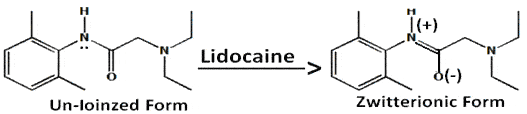
Insertion of a methylene group between the aromatic moiety and the carbonyl function in the procaine molecule, which prohibits the formation of the zwitterions, has with greatly reduced anesthetic potency. This observation lends further support for possible involvement of zwitterionic form when a local anesthetic binds to the receptor. A similar zitterionic form can also be envisioned in the amino amide series (ex:-Lidocaine).
Another important aspect of aromatic substitution- in the amino amides (lidocaine analogs), the o,o-di-methyl groups are required to provide suitable protection from amide hydrolysis.
Intermediate-chain:The Intermediate chain almost always contain a short alkylene chain of one to three carbons in length linked to the aromatic ring via several possible organic functional groups.
The nature of this intermediate chain determines chemical stability duration of action & relative toxicity. Placement of small alkyl groups (i.e., branching), especially around the ester function (hexylcaine & meprylcaine) or the amide function (etidocaine & prilocaine), also hinders ester or amide hydrolysis and prolongs the duration of action.
Legthening of the alkylene chain form one to two to three increases the pKa of the group from 7.7to 9.0 to 9.5. Thus, lengthening of the intermediate chain effectively reduces local anesthetic potency as a result of a reduction of onium ions under physiologic conditions.
Hydrophilic-Portion: Most clinically useful local anesthetics have a tertiary alkylamine, which readily forms water-soluble salts with mineral acids, and this portion is commonly considered as the hydrophilic portion of the molecule. It is often suggested that the tertiary amine function is needed only for the formation of water-soluble salts. However, it is quite conceivable that the onium ions produced by protonation of the tertiary amine group are also required for binding to the receptors.
Become a member of GPATTutor.com and access all the sections!
If you are a new user Register Now Existing User Login Now
